A 14 year-old female with Ehlers-Danlos Syndrome presenting for a shoulder reconstruction
By Alexandra Szabova, MD; John B. Rose, MD
Cincinnati Children’s Hospital Medical Center, Cincinnati, OH
Introduction
Ehlers-Danlos syndrome (EDS) is a group of hereditary connective tissue disorders. The clinical presentation of EDS is variable in terms of signs and symptoms as well as severity of illness and outcomes. In the majority of cases, inheritance is autosomal dominant. Several phenotypes exist including the most frequently encountered: classic, hypermobile, vascular and kyphoscoliosis types. The pathophysiology of pain in EDS is not fully understood. In most severe cases, the mechanism seems to be overlapping with other chronic musculoskeletal pain conditions, such as fibromyalgia. Not all patients with hypermobility have pain and not all patients with hypermobility and pain localize their pain in joints. The pain can be generalized, wide spread, out of proportion to objective findings, and disabling. The pain may also be associated with a variety of other troubling signs and symptoms including abdominal pain, nausea, constipation, fatigue, depression, anxiety, syncope, orthostatic intolerance, insomnia, migraine, diaphoresis, flushing, cold intolerance, and rash.
Case
A 14 year old, 45 kg female with Ehlers-Danlos Syndrome, multiple hypermobile joints, recurrent right shoulder dislocation, chronic shoulder pain, chronic diffuse musculoskeletal pain, insomnia, chronic fatigue, and generalized anxiety disorder presents for shoulder reconstruction.
The night before surgery, this patient took diazepam 5 mg to help her sleep. She also took oxycodone 5 mg at bedtime. The following morning she took her usual morning medications oxycodone 5 mg, gabapentin 600 mg and citalopram 20 mg PO with a sip of water at 6 AM. At 10 AM in the preoperative holding area, the patient’s intravenous catheter was inserted before entering the operating room without premedication.
After an uneventful intravenous induction of general anesthesia with fentanyl 100 mcg, propofol 200 mg, and rocuronium 40 mg, the patient’s trachea was intubated and she was placed in a beach chair position on the operating room table. Pressure points were carefully padded. Over three hours, general anesthesia was maintained with desflurane in oxygen and air and supplemented with additional intravenous medications including propofol 60 mg, morphine 5 mg, vecuronium 4 mg, dexamethasone 4 mg, and ondansetron 4 mg. At the conclusion of surgery, an interscalene block and catheter were placed with ultrasound guidance and Ropivacaine 0.2% 15 mL was administered without difficulties. After reversal of neuromuscular blockade, and return of spontaneous ventilation the patient’s trachea was extubated awake.
In the postanesthesia care unit, the patient received diazepam 5 mg IV for the complaint of feeling stiffness in her neck muscles. However, she had no complaints of pain. The peripheral nerve catheter infusion of ropivacaine 0.2 % was started at 4 mL/hour but later increased to 6 mL/hour for complaints of incisional pain. Patient was transferred to her hospital room one hour later. Postoperative pain management was carried out by the acute pain service. The patient was placed on diazepam 3 mg IV q4h prn, morphine IV 3.6 mg q2h prn, ketorolac 30 mg IV q6h, gabapentin 600 mg PO TID, citalopram 20 mg PO q day, and acetaminophen 1000 mg IV q6h. Patient had done well overnight. On POD 1, she was started on oxycodone 5 mg q4h PRN, and morphine was stopped. The interscalene catheter infusion was continued at 6 mL/hr. The remaining medications did not change. On POD 2, all medications were switched to oral formulations. Oxycodone was increased to 7.5 mg q4h around the clock. The infusion was switched to OnQ Pump, and patient was discharged home.
Following hospital discharge, a home care nurse came daily to assess the patient and removed the interscalene catheter on POD 5. The patient underwent her first session of physical therapy with the catheter in place and tolerated the therapy very well. Once the catheter was discontinued, the patient noticed mild increase in pain. Pain was controlled with oxycodone 7.5 mg q4h PRN. She was seen two weeks postop in the orthopedics and pain clinic offices and was noted to be doing well. The incision was healing well and there was no redness, warmth, swelling, or drainage. She continued to take oxycodone 2-3x/day, mainly for her PT sessions.
Discussion
Joint hypermobility (ability to painlessly perform maneuvers beyond the normal range of movement) has a high prevalence, affecting up to 10-30% of general population. Joint hypermobility syndrome (JHS) describes a disorder in which joint hypermobility and pain occur together, but without the risks for potentially life threating complications seen in some forms of EDS. EDS is a group of hereditary connective tissue disorders, generally inherited in an autosomally dominant fashion or occurring sporadically as a result of de novo mutations. Hallmarks of EDS include joint hypermobility, skin hyperextensibility, and tissue fragility but all organ systems can be involved and with some forms of EDS, there is a potential for life-threatening complications including spontaneous rupture of viscus organs or blood vessel dissection. Compared to JHS, the prevalence of EDS is low (1:5000). Incidence of mitral valve prolapse is 6% and aortic root dilatation 6 - 12% in patients with EDS.
There are six types of EDS (Villefranche classification), with classic, hypermobility and vascular types being most frequent. Joint hypermobility is a landmark symptom. Besides multiple joint injuries, dislocations, or subluxations, patients may present with poor wound healing, unusual scars, bruising, and musculoskeletal pain. Pes planus, pectus excavatum, spontaneous pneumothorax, myopia, muscle hypotonia, cervical insufficiency and uterine prolapse may be present in patients with EDS. Other comorbidities associated with EDS are fibromyalgia, functional abdominal pain (e.g. irritable bowel syndrome), fatigue, poor sleep, orthostatic intolerance, Postural Orthostatic Tachycardia Syndrome (POTS), headache, anxiety and depression. Joint laxity is assessed by Beighton hypermobility scale where greater than four out of nine points is diagnostic for hypermobility (Figure 1).
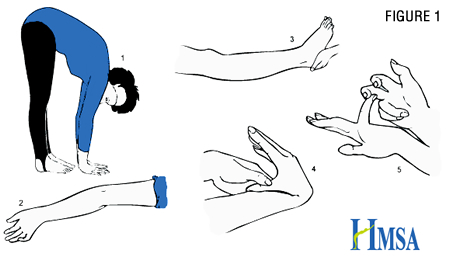
Patients may present early in age (usually more severe forms) or adolescence, following growth and hormonal changes. Joint instability and resulting injuries can be disabling. Surgical treatment is challenging due to poor wound healing leading to high risk for reinjury. Conservative treatment is crucial to prevent injuries and improve postsurgical outcomes. Patients with severe forms benefit from specialized centers. The focus of physical therapy is joint stabilization, strengthening and gentle stretching. Progression is slower than observed in normal patients following routine pediatric orthopedic surgery. NSAIDs are frequently utilized; muscle relaxants must be used with caution due to the risk of decreasing muscle tone and loss of joint protection. Opioids are rarely prescribed, with exception of most severe cases, and for acute exacerbations (trauma or surgery). Severe hypermobility may be a life changing condition and if not addressed aggressively, quite disabling. Patients need the support of psychologists in pain focused cognitive behavioral therapy, emphasizing good coping, mood and anxiety management. Athletic activities are encouraged in a “smart” way – avoiding contact sports and high impact activities like long distance running. Lifelong home exercise program is a must.
From the anesthesiologist’s perspective, connective tissue laxity affects the vascular system. Patients may present with orthostatic intolerance. In fact Postural Orthostatic Tachycardia Syndrome is common in some forms of EDS. Also, skin and joint laxity predisposes patients to a higher risk of intraoperative injuries due to positioning. Positioning needs to be meticulous - with extremity and joint stabilization in a neutral position and effective padding of skin and pressure points. Temporomandibular joint laxity may predispose some patients with EDS to jaw dislocation during routine airway management.
Peripheral nerve catheters have been utilized in children with chronic pain conditions to facilitate pain relief and physical therapy. We are gaining more experience in managing pediatric patients with peripheral nerve catheters in outpatient setting. We are also collecting data regarding safety and efficacy of catheter techniques in this population.
References
-
Pediatric Anesthesia 2010. 20: 318–322 - Perioperative anxiety in children
-
Pediatric Anesthesia 2009. 19: 1184–1190 - School-aged children’s experiences of postoperative music medicine on pain, distress, and anxiety
-
Can J Anaesth 2009. 56(11):843-50, Nov. Continuous peripheral nerve blocks for postoperative analgesia in children: feasibility and side effects in a cohort study of 339 catheters.
-
Clin Genet 2012. 82: 1–11. The Ehlers–Danlos syndrome, a disorder with many faces.
-
Rheum Dis Clin N Am 2013. 39: 419-430. Joint Hypermobility Syndrome.

